ISO Changes: Managing Risk
The new ISO 9001:2015 standard emphasizes risk assessment. Medical shops with ISO 13485 certification already manage this.
Share





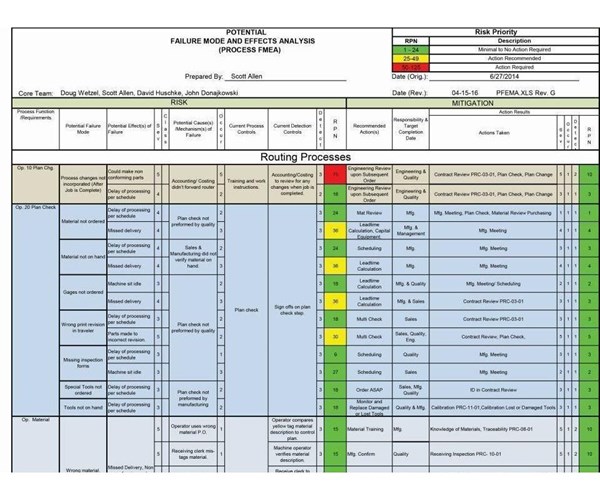
This is just a small portion of the process failure mode and effects analysis (PFMEA) document that Protomatic developed. It identifies more than 300 potential failure modes and offers procedures to mitigate those risks.
After the end of September 2018, a certificate to ISO 9001:2008 will no longer be valid. One change in its replacement ISO 9001:2015 is a requirement to establish procedures for comprehensive risk management. Shops with medical ISO 13485 certification already manage this.
Failure mode and effects analysis (FMEA) is one common method of risk identification and mitigation, although it is sometimes referred to differently from company to company. Protomatic, a Dexter, Michigan, prototype machining and custom short-run production facility serving the medical, automotive, military and aerospace industries, refers to its as PFMEA—process failure mode and effects analysis.
Learning about the risk management procedures this shop has put in place might serve as a good model for other, non-medical shops that will be working toward becoming registered to the updated standard. Learn more.
Related Content
-
Ametek EMC Laser Cutting System Offers Through-Part Cooling
The new laser cutting system offers comprehensive Swiss machining capabilities, as well as through-part cooling and automatic part-handling options.
-
Kennametal Turning Tools Enhance Small Part, Medical Machining
TopSwiss Micro Boring Solid tooling and KCU25B turning inserts are well suited for the small parts and medical machining space.
-
Mikron Milling Tool Suppresses Machining Chatter
IMTS 2024: The CrazyMill Cool CF milling tool features chatter suppression during machining, especially on thin-walled parts, which enables smoother cutting.


















.jpg;maxWidth=300;quality=90)


